Datt Mediproducts (DMPL) incorporated as a Limited Liability Company with its Registered Office in the state of West Bengal, India
OUR COMPANY JOURNEY
-
 1995, January
1995, January -
 1996, November
1996, NovemberDMPL completes construction of its factory and installation of imported plant & machinery and other world class infrastructure at Sohna, Haryana, India
-
 1997, February
1997, FebruaryDMPL commences commercial production of all its medical devices
DMPL sets up its first Class 100,000 Clean Room at its manufacturing facility as per International standards/norms
-
 1998, March
1998, MarchDMPL obtains International certification - ISO, CE, from SGS Yarsley, United Kingdom
DMPL starts exports of its medical devices to UK, Germany, Spain, France, etc.
-
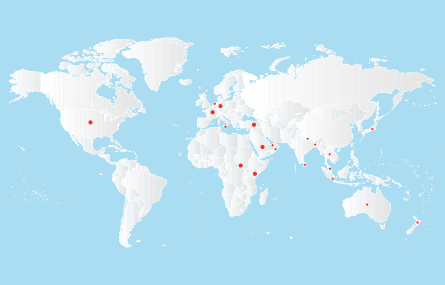 2001, January
2001, JanuaryDMPL expands its International reach to Australia, New Zealand, South Africa, Japan, UAE and a number of countries in Europe
-
 2007, October
2007, OctoberDMPL commences its Indian sales operations and creates its own unique brand logo and identity for its indigenously produced devices
-
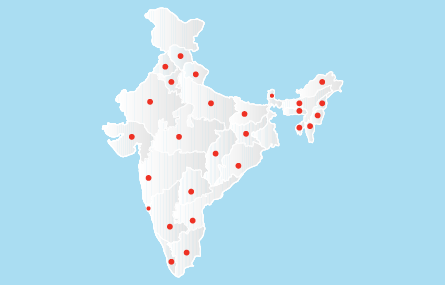
 2008, January
2008, JanuaryDMPL establishes a complete distribution network in India by appointing distributors, C&F agents and sales staff in all Indian states with a total workforce of more than 100 people
-
 2009
2009DMPL forms a specialized “Department of Life Sciences” and appoints bio-technologists and medical doctors ?? to research and develop economical advanced medical devices to cater to the requirement of the deprived segment of India’s population who cannot afford much needed expensive imported medical devices
-
 2011, November
2011, NovemberDr. Rajan Datt, Managing Director and DMPL awarded by the Textiles Institute, Manchester, UK for New Material Award in medical devices.
-
 2014
2014DMPL obtains registration with Department of Scientific and Industrial Research (DSIR) for its R&D Laboratory to research and develop niche advanced wound care devices along with registration under Section 35(2AB) of the Income Tax Act
-
 2014-2018
2014-2018DMPL files for patents for its unique developments in advanced medical devices/processes.
Hemostat (Velseal T and Velseal), VelVert, Velnez, Velgraft , Velgraft amended Claims First and Velgraft amended Claims First
-
 2018-2019
2018-2019DMPL is awarded 6 patents by the US Patent
Hemostat (Velseal T and Velseal), VelVert, Velnez, Velgraft , Velgraft amended Claims First and Velgraft amended Claims First